Macroscopic Characteristics & Bond Interactions
Physical Processes and Intermolecular Interactions
- During physical processes, the chemical composition of a substance does not change
- However, there is a change in the appearance of the substance
- This change in appearance involves changes in the intermolecular forces between the molecules or ions that are part of the compound
- E.g. When water turns into steam, hydrogen bonding which is an intermolecular interaction between water molecules, needs to be broken down before changing state
- The polar covalent bonds between oxygen and hydrogen atoms remain the same because the internal composition of water does not change when going from liquid to gas
- All the changes of state involve the breaking of intermolecular interactions such as: hydrogen bonding, dipole-dipole, and London dispersion forces
Hydrogen bonding interaction and polar covalent bonds in water

Intermolecular interactions are broken when going from liquid to gas. Intramolecular interactions remain the same
Chemical Processes and Bond Breaking/Formation
- During chemical processes, the chemical composition of substances changes
- Reactants transform into products whose internal structure is totally different because the bonds between atoms rearrange
- Chemical processes are a game that involves breaking and forming chemical bonds
- Bond breaking is a process that requires energy
- Bond formation is a process that releases energy
- All the chemical reactions are chemical processes because the chemical identity of the products is different than the identity of the reactants
- E.g. When hydrogen gas is burned in the presence of oxygen gas, water is produced
- In the reactants, there are two H–H bonds and one O=O bond. While in the products there are four O–H bonds
- There is a rearrangement of the covalent bonds by comparing the left side with the right side
Burning hydrogen gas with oxygen gas to produce water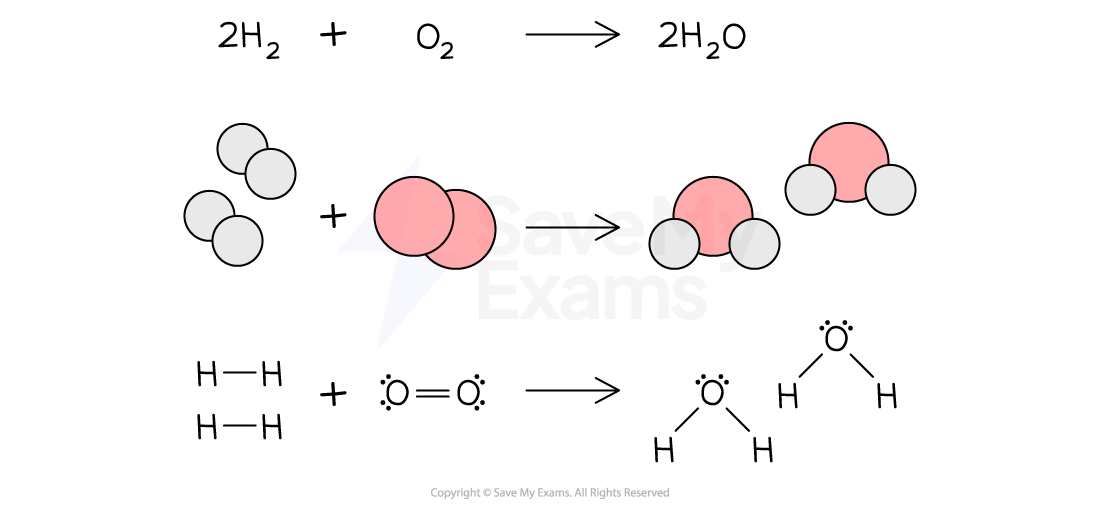
Chemical reaction between hydrogen and oxygen to produce water by showing the covalent bonds that were broken and formed during the process


