Atomic Structure
The Atom
- The atom is the smallest building block of matter
- It consists of three subatomic particles
- Protons are positive particles found in the nucleus
- Neutrons are neutral particles found in the nucleus
- Electrons are negative particles that orbit the nucleus in electron shells
- The electrons determine the chemical properties of the element
- Neutral atoms have the same amount of protons and electrons
Atomic Number and Mass Number
- The atomic number (Z) of an element is the number of protons in its nucleus
- It defines the element's identity and it always a whole number in the periodic table
- Z = Number of protons
- It defines the element's identity and it always a whole number in the periodic table
- The mass number (A) is the sum of protons and neutrons in the nucleus
- It represents the atom's mass
- Electrons do not contribute to the mass of the atom because they are too light compared with protons or neutrons
- A = Number of protons + Number of neutrons
- Isotopes are atoms of the same element with the same atomic number but different mass numbers. Therefore, protons are the same but neutrons are different
- Elements are usually represented with the A/Z notation
The beryllium atom
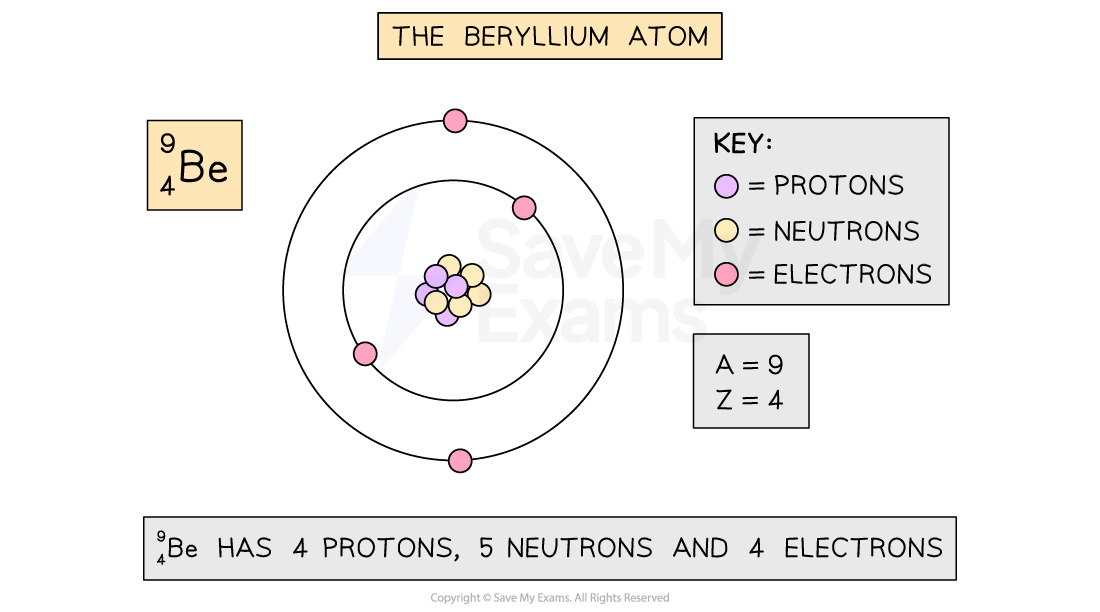
The beryllium atom contains 4 protons, 5 neutrons and 4 electrons
Worked example
Calculate the atomic number and mass number for an atom with 17 protons and 20 neutrons. After this, identify the element and write it down using the A/Z notation.
Answer
- Step 1: Identify the given information
- The problem provides the number of protons (17) and neutrons (20).
- Step 2: Determine the atomic number (Z)
- The atomic number (Z) represents the number of protons in an atom. It also defines the element's identity.
- Z = 17
- So, the atomic number is 17, which corresponds to the element chlorine (Cl) on the periodic table.
- Step 3: Calculate the mass number (A)
- The mass number (A) is the sum of protons and neutrons in the nucleus.
- A = Number of protons + Number of neutrons
- A = 17 protons + 20 neutrons
- A = 37
- The mass number (A) for this atom is 37.
- Step 4: Identify the element
- With an atomic number of 17 (Z = 17), we can determine that the element is chlorine (Cl).
- So, the atom in question is a chlorine atom with 17 protons, 20 neutrons, an atomic number (Z) of 17, and a mass number (A) of 37.
- Step 5: Write it down with the A/Z notation
The Rutherford Experiment
- Rutherford performed an experiment to understand the structure of the atom
- He bombarded a gold foil with tiny positive particles, called alpha particles
- Most alpha particles passed through the foil, but some were significantly deflected, and a few of them bounced back
- Since particles with the same charge repel, Rutherford concluded that the atoms should contain an small, dense, and positively charged core at the center
- This core was named nucleus
- The nucleus contain protons and neutrons
The Rutherford Experiment
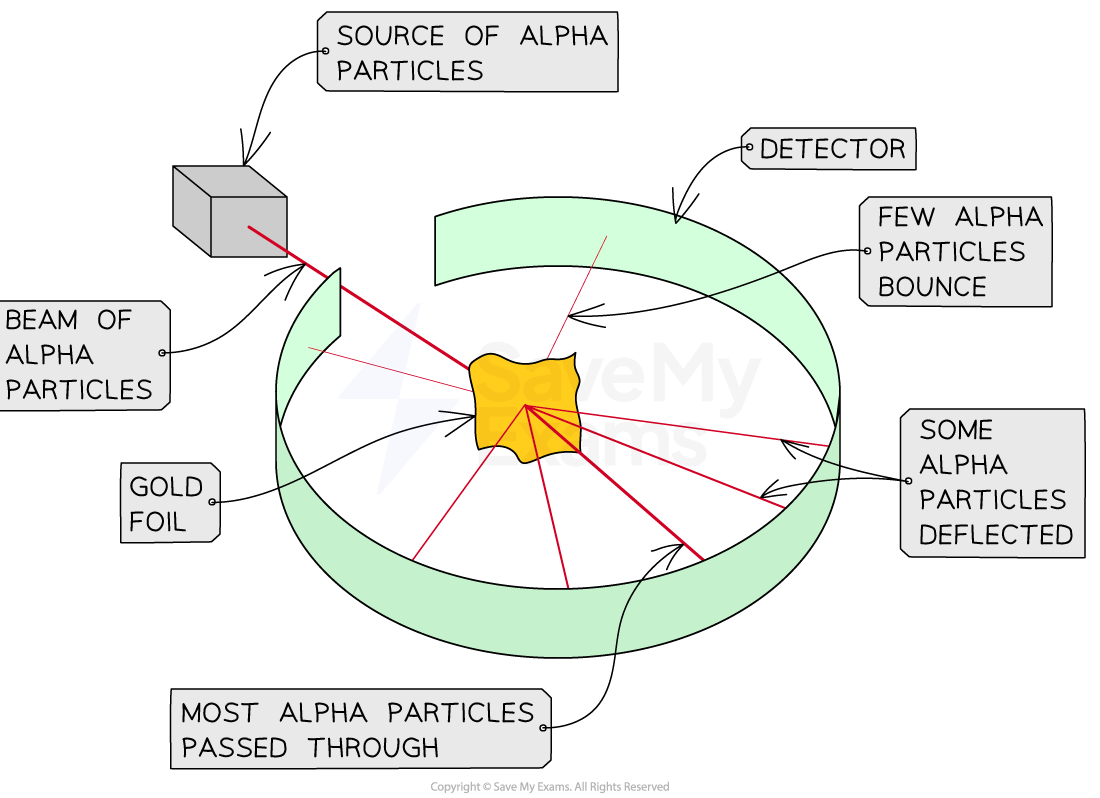
The Rutherford experiment showed atoms contain a small, dense, and positively charged core at the center

