Solubility of Ionic & Molecular Compounds
- The solubility of two substances depends on their intermolecular interactions
- Ions and molecules can exhibit can exhibit the following interactions:
Relative strength comparison between the intermolecular interactions
|
Interaction |
Relative strength |
|
Ion-dipole |
Strongest |
|
Hydrogen bonding |
|
|
Dipole-dipole |
|
|
London dispersion forces |
Weakest |
- A solution is made from two components a solvent and a solute
- In a solution system, intermolecular interactions exists in three possible scenarios:
- Solvent-solute, i.e solvent particles with solute particles
- Solvent-solvent, i.e. solvent particles with themselves
- Solute-solute, i.e.solute particles with themselves
When does a solution form?
- A solution is formed when solvent-solvent interactions and the solute-solute interactions are the same
- When this happens, the substances are said to be miscible
- E.g. Ethanol and water present hydrogen bonding interactions. Therefore, they are miscible in one another
- When the solvent-solvent interaction and the solute-solute interaction are not equal, the strength of the solvent-solute interaction must be considered
The three interactions between the particles of solute and solvent

Solvent-solvent, solvent-solute and solute-solute interactions must be considered for solubility
- A solution is formed when the solvent-solute interactions are stronger than the solvent-solvent interactions
- E.g. Salt and water present ion-dipole interactions. Water molecules present hydrogen bonding between themselves
ion-dipole > hydrogen bonding
-
- Therefore, they are miscible in one another
- A solution will not be formed when the solvent-solute interactions are weaker than the solvent-solvent interactions
- E.g. Hexane and water present London Dispersion Forces. Water molecules present hydrogen bonding themselves
London-Dispersion Forces < hydrogen bonding
-
- Therefore, they are not miscible in one another
- The rules of solution formation are summarized in the table below
Rules of Solution Formation by comparing the intermolecular interactions strengths
|
Comparison of the strength between intermolecular interactions |
Solution formed? |
|
Solvent-solute = Solvent-solvent |
Yes |
|
Solvent-solute > Solvent-solvent |
Yes |
|
Solvent-solute < Solvent-solvent |
May or may not form |
Worked example
Predict if a solution will be formed when chloroform is mixed with an acetone solvent.

Answer:
Step 1 (Optional): Draw the molecular structure of the solvent molecules and the solute molecules. This step is not applicable to ions since they exist as lattice structures
Step 2: Identify the solvent-solvent intermolecular interactions
- Acetone is our solvent. Since oxygen is more electronegative than carbon, a dipole moment will be formed
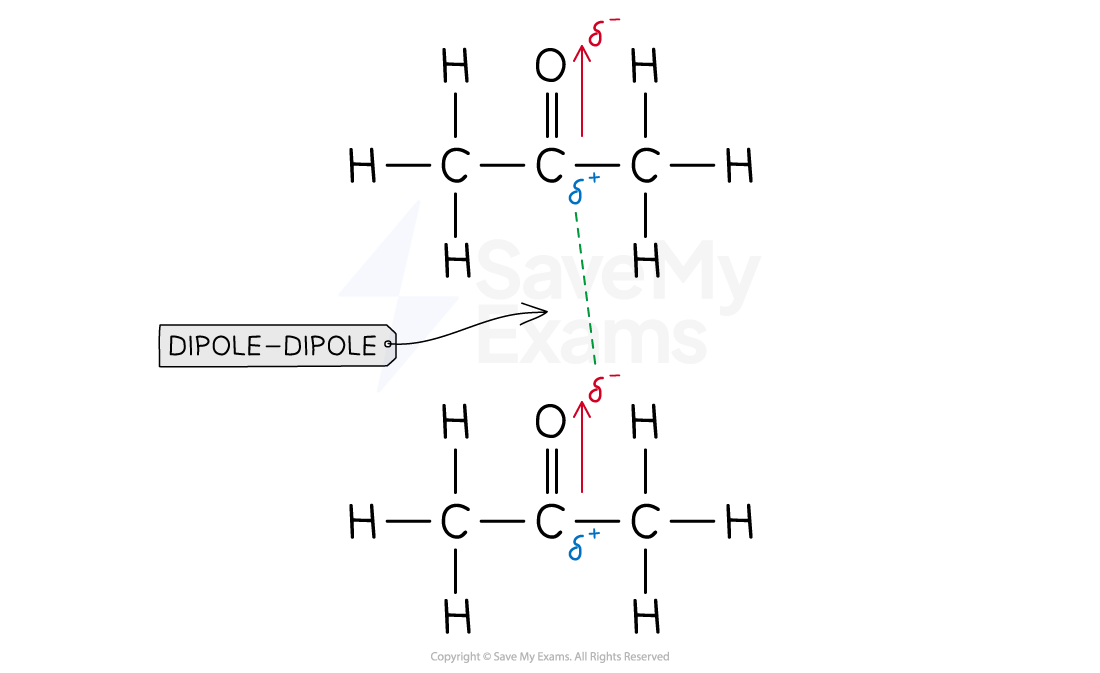
- Therefore, the solvent-solvent intermolecular interaction is dipole-dipole
Step 3: Identify the solute-solute intermolecular interactions
- Chloroform is our solute. Since it is a non-symmetrical molecule and chlorine is more electronegative than carbon, dipole moments will be formed

- Therefore, the solute-solute intermolecular interaction is dipole-dipole
Step 4: If the solute-solute interaction and the solvent-solvent interactions are the same, they are miscible. Otherwise, the table should be used and the solvent-solute interactions must be considered
- Since acetone presents dipole-dipole interactions and chloroform presents dipole-dipole interactions, both solute and solvent will be miscible in one another
Worked example
Predict if a solution will be formed when hexane is mixed with water as solvent.

Answer:
Step 1 (Optional): Draw the molecular structure of the solvent molecules and the solute molecules. This step is not applicable to ions since they exist as lattice structures
Step 2: Identify the solvent-solvent intermolecular interactions
- Water is our solvent. Since hydrogen is bonded to a highly electronegative atom such as oxygen, a strong dipole moment will be formed
- Therefore, the solvent-solvent intermolecular interaction is hydrogen bonding

Step 3: Identify the solute-solute intermolecular interactions
- Hexane is our solute. Since hexane is a symmetrical molecule, all its dipole moments are canceled out
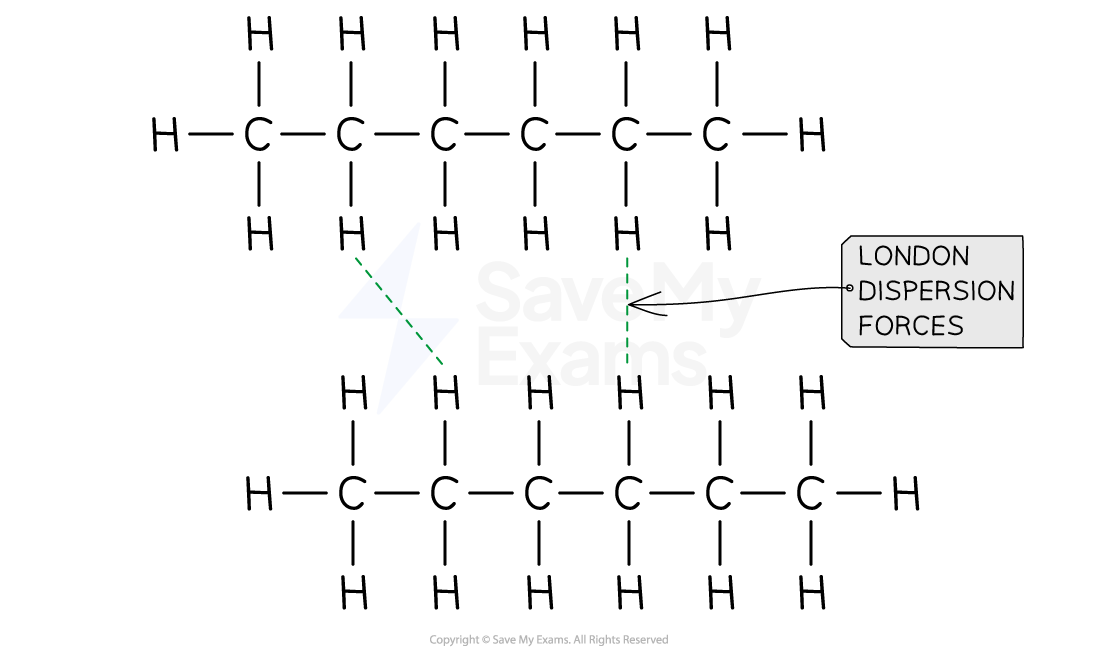
- Therefore, the solute-solute intermolecular interaction are London dispersion forces
Step 4: If the solute-solute interaction and the solvent-solvent interactions are the same, they are miscible. Otherwise, the table should be used and the solvent-solute interactions must be considered
- The solvent-solvent interaction is not the same as solute-solute interaction
- The solvent-solute interaction must be considered
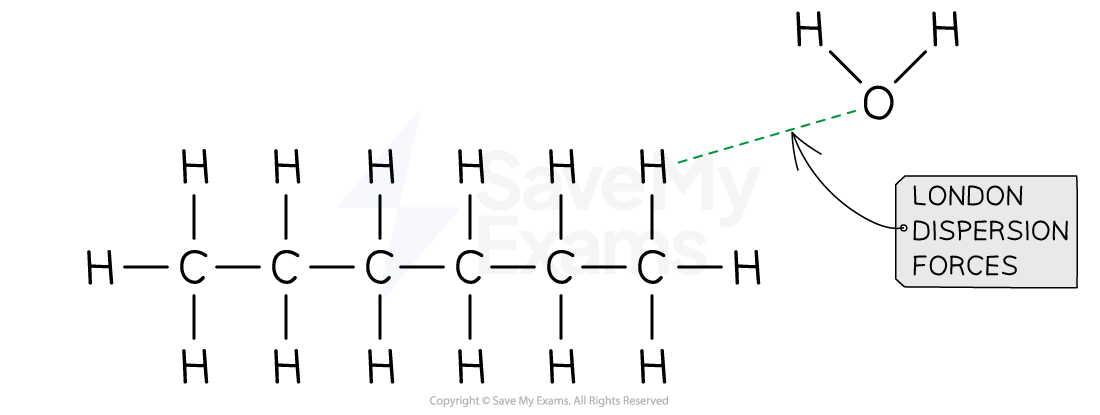
- As shown in the image below, hexane and water present London dispersion forces interactions because oxygen induces momentaneous dipoles in hexane
- Using the rules of solution, if solvent-solute < solvent-solvent, a solution will not be formed
- Since London-dispersion forces < hydrogen bonding in terms of relative strength, hexane and water are not miscible, and a solution will not be formed

