A PES analysis of an unknown element (X) was performed and the spectrum below was obtained. The sample does not contain any impurities. What is the identity of X?

Unknown sample PES Spectrum
Answer:
- Step 1: Analyze the number of peaks
- Since there are three peaks, this will correspond to the three first subshells of an atom: 1s, 2s and 2p
- Step 2: Analyze the position of the peaks
- The peak with the highest binding energy will correspond to the 1s subshell since it is the closest to the nucleus, and the coulombic attractions are stronger
- The peak with the lowest binding energy will correspond to the 2p subshell since it is the farthest to the nucleus, and the coulombic attractions are weaker
- Therefore, the peak in the middle will correspond to the 2s subshell
- Step 3: Analyze the height of the peaks
- When comparing the height of the three peaks, they are all the same.
- Therefore, the same number of electrons have been removed in the 1s, 2s and 2p
- Since, subshells 1s and 2s can hold up to 2 electrons, the amount of electrons in the 2p subshell for this atom was 2
- Step 4: Predict the electron configuration of the atom
- 1s2 2s2 2p2
- We can connect the electron configuration with the PES Spectrum labeled below
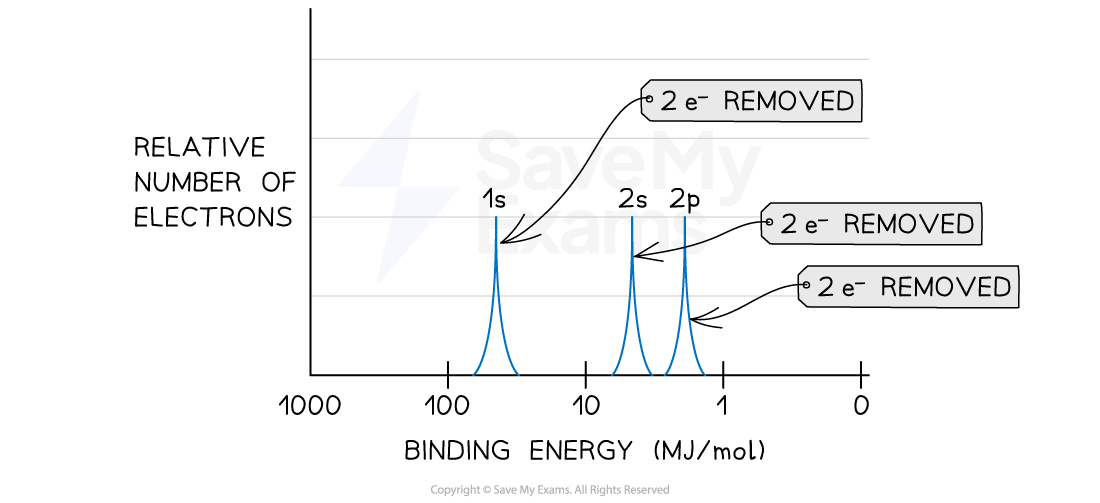
PES Spectrum labeled
- Step 5: Identify the element using the periodic table
- There are two ways to identify the unknown element:
- Counting the electrons and calculating the protons
- 6 electrons and, since it was a neutral sample, there are 6 protons
OR
-
-
- Counting the valence electrons and identify the period
- There are 4 valence electrons, therefore the element should be in Group 14 of your AP Chemistry Periodic Table
- Since the valence electrons are in the second shell, the element should be in the second period of the periodic table
- Therefore, element X is carbon (C)



