Cell Potential & Equilibrium
Deviations from standard conditions and cell potential
- Equilibrium arguments, such as Le Châtelier’s principle, do not apply to electrochemical systems because these systems are not in equilibrium
- For a standard cell where the concentrations are the same, the reaction quotient, Q, will be equal to 1
Reaction quotient, Q =
- Since we know that the standard cell potential is calculated at 1 M and 1 atm, we know that the value for Q (the reaction quotient) would have to be 1
- As a cell reaches equilibrium, the reaction quotient reaches the equilibrium constant, K, (Q = K ) and the magnitude of the cell potential decreases to zero
- The cell is not creating a voltage and is now 'dead'
- A value for the cell potential of zero means that the reaction has reached equilibrium
- If we change the concentration, we can change the magnitude of Ecell
- This is now no longer a cell operating under standard conditions!
Changing concentrations in a galvanic cell

Standard conditions in a Zn/Zn2+ Cu2+/Cu cell
| Zn (s) → Zn2+ (aq) + 2e– | Eo = -0.76 V | Oxidation |
| Cu2+ + 2e– → Cu (s) | Eo = +0.34 V | Reduction |
Eo cell = Eored - Eoox
Eocell = 0.34 - (-0.76) = 1.10 V
- If we change the concentrations to:
| New Concentration | New Concentration |
|
[Zn2+] = 5.00 M [Cu2+] = 1.00 M |
[Zn2+] = 1.00 M [Cu2+] = 5.00 M |
|
Q = 5 |
Q = 0.2 |
|
|
|
|
Ecell = 1.10 - 0.02 = 1.08 V which is now a lower value |
Ecell = 1.10 - (-0.02) = 1.12 V which is now a higher value |
|
This is a deviation that takes the cell further from equilibrium than Q = 1 will increase the magnitude of the cell potential relative to Eocell |
This is a deviation that takes the cell closer to equilibrium than Q = 1 will decrease the magnitude of the cell potential relative to Eocell |
Concentration Cells
- A voltage can be generated by constructing an electrochemical cell in which each compartment contains the same redox active solution but at different concentrations
- The voltage is produced as the concentrations equilibrate
- For example, if we look at the following cell containing two different concentrations of zinc sulfate solution
Diagram to show two different concentrations of zinc sulfate in a concentration cell
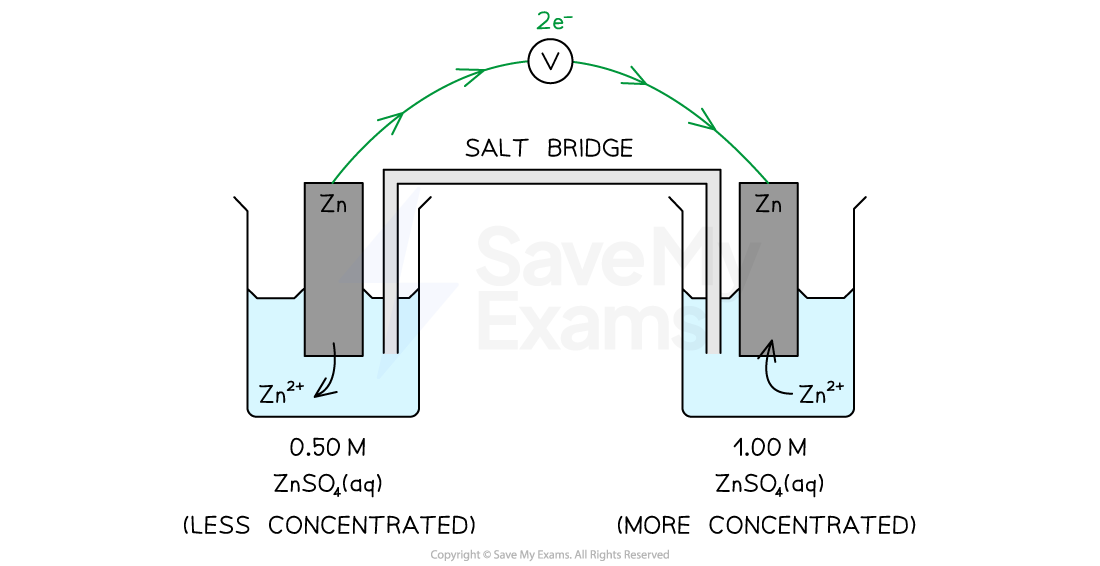
Reduction occurs at the more concentrated half-cell and oxidation occurs at the less concentrated half-cell
- The two different reactions taking place are
- Oxidation occurs in the less concentrated half-cell:
- Zn2+ (aq) (0.50 M) + 2e– → Zn (s)
- Reduction occurs in the more concentrated half-cell:
- Zn (s) → Zn2+ (aq) (1.00 M) + 2e–
- Oxidation occurs in the less concentrated half-cell:
- So for this reaction, Ecell will = 0
- Eocell = Eored - Eoox
- Eocell = -0.76 - (-0.76) = 0.00 V
- From the concentrations of the two different solutions of zinc sulfate, we can calculate the value for Q, or the reaction quotient
- Q =
- In this example Q =
- Solids are not included in this calculation
- Q =
- Therefore, Ecell =
- Ecell = 8.91 x 10-3 V
- This is positive so is a spontaneous reaction
- As the concentrations of each half cell get closer together over time, the value for Q will increase and Ecell will decrease
- When Q = 1 the reaction will be in equilibrium and Ecell will be equal to 0
- If there is no difference in concentration, there is no longer a requirement for concentrations to equalise which creates the potential difference in the cell
- If Ecell = 0, then the cell is 'dead'

