Volumetric Analysis
REQUIRED PRACTICAL 1
Performing the Titration
- The key piece of equipment used in the titration is the burette
- Burettes are usually marked to a precision of 0.10 cm3
- Since they are analogue instruments, the uncertainty is recorded to half the smallest marking, in other words to ±0.05 cm3
- The end point or equivalence point occurs when the two solutions have reacted completely and is shown with the use of an indicator

The steps in a titration
- A white tile is placed under the conical flask while the titration is performed, to make it easier to see the colour change

- The steps in a titration are:
- Measuring a known volume (usually 20 or 25 cm3) of one of the solutions with a volumetric pipette and placing it into a conical flask
- The other solution is placed in the burette
- To start with, the burette will usually be filled to 0.00 cm3
- A few drops of the indicator are added to the solution in the conical flask
- The tap on the burette is carefully opened and the solution added, portion by portion, to the conical flask until the indicator starts to change colour
- As you start getting near to the end point, the flow of the burette should be slowed right down so that the solution is added dropwise
- You should be able to close the tap on the burette after one drop has caused the colour change
- Multiple runs are carried out until concordant results are obtained
- Concordant results are within 0.1 cm3 of each other
Recording and processing titration results
- Both the initial and final burette readings should be recorded and shown to a precision of ±0.05 cm3, the same as the uncertainty
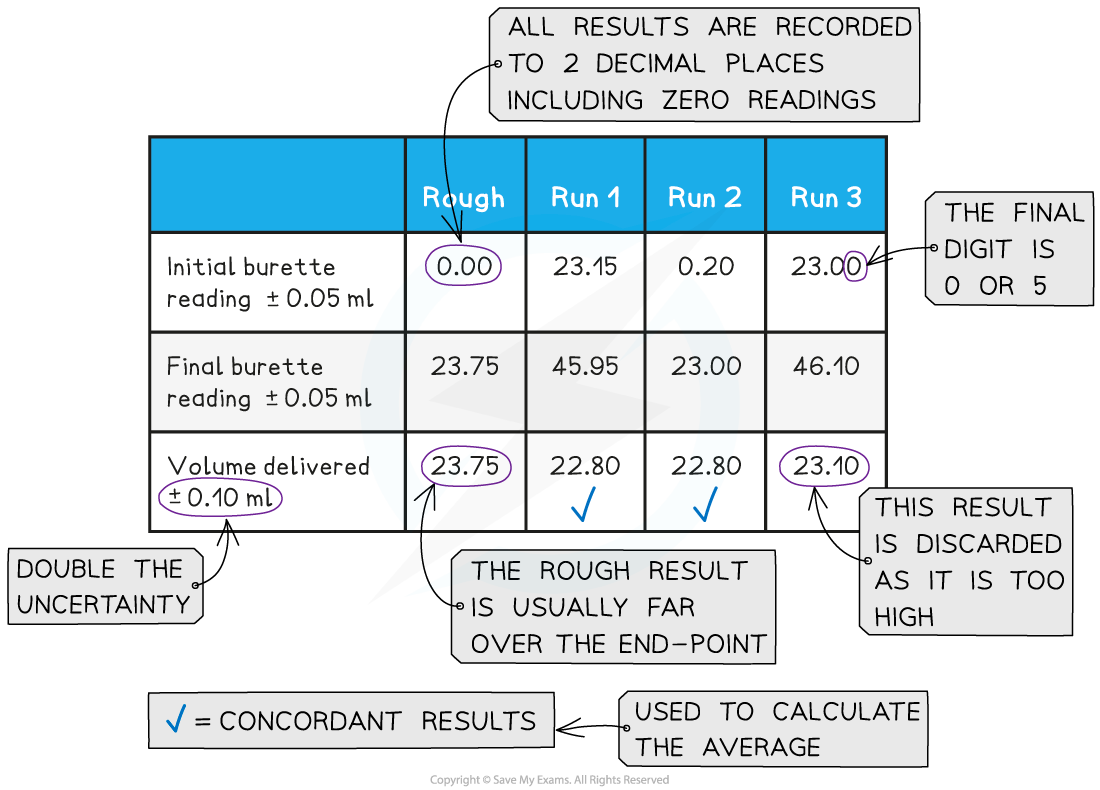
A typical layout and set of titration results
- The volume delivered (titre) is calculated and recorded to an uncertainty of ±0.10 cm3
- The uncertainty is doubled, because two burette readings are made to obtain the titre (V final – V initial), following the rules for propagation of uncertainties
- Concordant results are then averaged, and non-concordant results are discarded
- The appropriate calculations are then done
Percentage Uncertainties
- Percentage uncertainties are a way to compare the significance of an absolute uncertainty on a measurement
- This is not to be confused with percentage error, which is a comparison of a result to a literature value
- The formula for calculating percentage uncertainty is as follows:
 Adding or subtracting measurements
Adding or subtracting measurements
- When you are adding or subtracting two measurements then you add together the absolute measurement uncertainties
- For example,
- Using a balance to measure the initial and final mass of a container
- Using a thermometer for the measurement of the temperature at the start and the end
- Using a burette to find the initial reading and final reading
- In all these example you have to read the instrument twice to obtain the quantity
- If each you time you read the instrument the measurement is ‘out’ by the stated uncertainty, then your final quantity is potentially ‘out’ by twice the uncertainty

