Variable Oxidation States
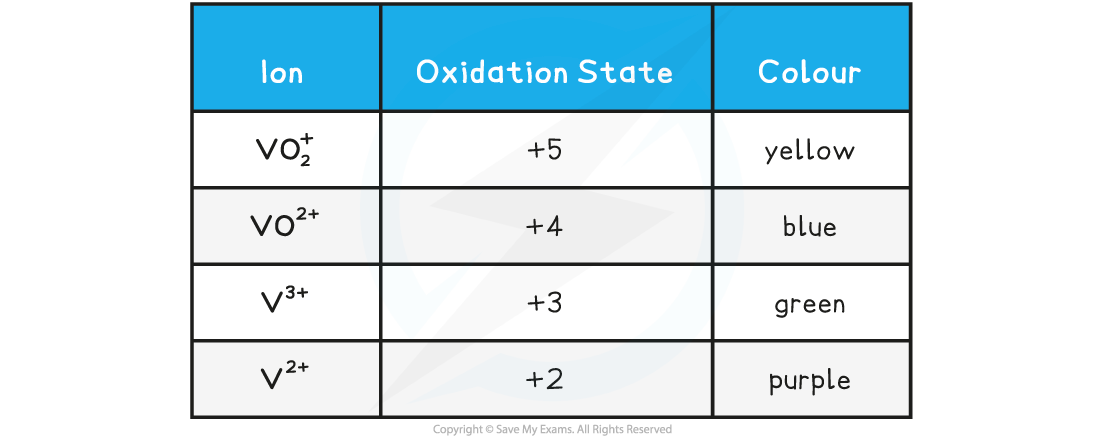
- The table below shows the most common oxidation states of a few transition metals
Oxidation states of transition elements table

- When transition elements forms ions they lose electrons from the 4s subshell first
- This is because when the orbitals are occupied, the repulsion between electrons pushes the 4s into a higher energy state so that it now becomes slightly higher in energy than the 3d subshell
- The 4s is now the outer shell and loses electrons first
- The loss of the 4s electrons means that +2 is common oxidation state in transition metals
- The reason why the transition metals have variable oxidation states all comes down to energy

Ionisation energies for the removal of successive electrons in titanium and vanadium
- We can see from the graph that the first few ionisation energies are relatively small and relatively close together
- This means that the energy difference associated with removing a small number of electrons enables transition metals to vary their oxidation state with ease